Eli Lilly & Co. had its patent on Zyprexa®, the world’s top- selling schizophrenia drug, upheld by the Court of Appeals for the Federal Circuit. The court affirmed a lower court decision that the patent is valid stating that for a chemical compound, a prima facie case of obviousness requires “structural similarity between claimed and prior art subject matter . . . where the prior art gives reason or motivation to make the claimed compositions.” Eli Lilly v. Zenith Goldline Pharmaceuticals, Inc. and Teva and Dr. Reddy’s (05-1396, -1429, -1430).
Lilly had sued for infringement of U.S. Pat. No. 5,229,382 after Zenith Goldline (now IVAX), Dr. Reddy’s Laboratories, and Teva Pharmaceuticals filed an Abbreviated New Drug Application (ANDA). The U.S. District Court for the Southern District of Indiana found the ’382 patent valid and infringed and the Appeals Court affirmed.
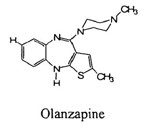
In seeking to have the patent declared invalid based on obviousness, IVAX argued that the district court erred by erecting “a threshold requirement that defendants establish a teaching or incentive to treat the closest prior art (i.e., Compound ‘222) as a ‘lead compound.’” IVAX also claimed that the district court disregarded (1) the structural characteristic of olanzapine as the adjacent homolog of Compound ‘222, (2) the suggestions to delete fluorine from the prior art compound flumezapine, and (3) the observation that Compound ‘222 and flumezapine “bracket” olanzapine.
The CAFC avoided an issue with the KSR “teaching-suggestion-motivation” test stating that:
The factual underpinnings are: (1) the scope and content of the prior art, (2) the differences between the prior art and the claimed invention at the time of invention, (3) the level of ordinary skill in the art, and (4) the objective indicia of nonobviousness. See Graham v. John Deere Co., 383 U.S. 1, 17 (1966); Panduit Corp. v. Dennison Mfg., 810 F.2d 1561, 1566-67 (Fed. Cir. 1987). For a chemical compound, a prima facie case of obviousness requires “structural similarity between claimed and prior art subject matter . . . where the prior art gives reason or motivation to make the claimed compositions.” In re Dillon, 919 F.2d 688, 692 (Fed. Cir. 1990) (en banc). “[A] reasonable expectation of success, not absolute predictability” supports a conclusion of obviousness. In re Longi, 759 F.2d 887, 896 (Fed. Cir. 1985).
The CAFC held that the reference did not suggest or make obvious, among other things, olanzapine’s hydrogen component. The prior art references at the time of this invention taught away from using a non-halogenated compound as a substituent in the benzene ring, exactly where olanzapine has a hydrogen atom.
Also, the CAFC did not feel that IVAX proved that a person ordinarily skilled in this art would have selected Compound ‘222 as a lead compound because it contained hydrogen rather than fluorine or chlorine noting that at the time of invention, the state of the art would have directed the person of ordinary skill in the art away from unfluorinated compounds like Compound ‘222:
After all, the primary example of the state of the art at that time, the ’574 patent, did not provide any biological data for compound ‘222, suggested a preference for halogen-containing compounds, and identified a fluorine-containing compound, ethyl flumezapine, as “particularly active.” Findings of Fact and Conclusions of Law, slip op. at p. 170. Moreover, as the trial court detailed, Chakrabarti 1980a expressly taught that the addition of a fluorine or chlorine enhanced anti-psychotic activity. It also taught that the unfluorinated Compound ‘222 was less active than the benchmark compound, clozapine. Id. Thus, rather than providing the requisite motivation, the prior art taught away from selecting Compound ‘222 as a lead compound for further development.
Furthermore, the court felt that Lilly overcame any prima facie case of obviousness with extensive secondary considerations to rebut obviousness such as (1) a long-felt and unmet need; (2) failure of others; (3) industry acclaim; and (4) unexpected results.
The CAFC also looked at the trial court’s conclusion that Lilly’s clinical trials of olanzapine were not a public, but an experimental, use that negated any section 102 bar. Under section 102, a person is entitled to a patent, unless “the invention was . . . in public use . . . in this country, more than one year prior to the date of the application for patent in the United States.” 35 U.S.C. § 102(b).
In considering whether a particular use was “public” within the meaning of section 102(b), the CAFC held that several indicia may show the negating experimental character of a use, including (1) the length of the test period, (2) any confidentiality agreement, (3) any records of testing, (4) any monitoring and control of the test results, (5) the number of tests, and (6) the length of the test period in relation to tests of similar inventions. In this case, Lilly restricted access to the facility and closely monitored and confined the movements of the volunteers.
Related:

[…] CAFC: You Still Need Motivation to Render Drug Patent ObviousPatent Baristas, OH – 18 hours agoEli Lilly & Co. had … drug. Eli Lilly v. Zenith Goldline Pharmaceuticals, Inc. and Teva and Dr. Reddy s (05-1396, -1429, -1430). … […]