Cordis and Johnson & Johnson tried to get a new trial and judgment as a matter of law (JMOL) following a jury verdict finding that they infringed Boston Scientific’s patent. Boston Scientific v. Cordis Corp. (08-1073). The U.S. Court of Appeals for the Federal Circuit said that “because the court erred … in failing to hold the ’536 patent to have been obvious, we reverse the judgment.”
Cordis makes the Cypher drug-eluting expandable stent, which has two coatings: an undercoat containing the drug and a topcoat. Early in the manufacturing process, the Cypher stent’s topcoat is drug-free. The topcoat is sprayed on as a solution containing no drug but containing a polymer and two solvents. The solvents dissolve the drug, allowing it to diffuse from the undercoat into the topcoat. Thus, when the Cypher stent is sterilized, removing any remaining solvent, the drug has moved into the topcoat to such an extent that the topcoat and undercoat contain the same concentration of drug.
U.S. Patent 6,120,536 relates to a drug-eluting expandable stent with a coating that has a non-thrombogenic surface. Boston Scientific sued Cordis alleging that Cordis’s Cypher stent infringed claim 8 of the ’536 patent.
The patent covers:
[An expandable stent comprising a tubular body having open ends and an open lattice sidewall structure] wherein at least a part of the device portion is metallic and at least part of the metallic device portion is covered with a coating for release of at least one biologically active material, wherein said coating comprises an undercoat comprising a hydrophobic elastomeric material incorporating an amount of biologically active material [and] a topcoat … comprising a biostable, non-thrombogenic material which provides long term non-thromobogenicity to the device portion during and after release of the biologically active material, and wherein said topcoat is substantially free of an elutable material [and wherein the coating conforms to said sidewall structure in a manner that preserves said open lattice].
The district court construed the phrase “non-thrombogenic material which provides long term non-thrombogenicity to the device portion during and after release of the biologically active material” in claim 1 as meaning “a material that does not promote thrombosis for a period of time that extends both during and after release of the biologically active material.” The court relied on medical dictionary definitions for the meaning of “thrombogenic” (“causing thrombosis or coagulation of the blood”) and “thrombolytic” (“break[ing] up or dissolv[ing] a thrombus”).
The court rejected Cordis’s proposed definition of “non-thrombogenic,” which required a “significant reduction in thrombogenicity over that experienced with bare metal stents.” The court also construed the phrase “substantially free of an elutable material” to mean “largely or approximately free of an elutable material.”
The court reasoned that Domb, which discloses esophageal stents, does not suggest the use of metal in a stent, and that there was no evidence of motivation to combine Domb with other references. The court reasoned that Wolff does not teach a metallic stent having a two-layer coating, and the failure of Wolff’s assignee to create the claimed stent after more than a decade of work evidenced a lack of motivation to combine the features of its various prior art stents with each other.
The court found support for the jury’s infringement finding in a witness’s testimony that the topcoat is drug-free when applied and “has about 1 to 2% drug . . . after manufacturing is completed,” finding that 1 to 2% drug may be considered “substantially free.” The court also pointed to evidence that a stent need not be sterilized to be implantable and thus that the stent could infringe during manufacture, when the topcoat was first applied and was then drug-free.
Finally, the court found that the ’536 patent specifically contemplates that the drug will move into the topcoat prior to implantation, implying that the “substantially free” limitation does not preclude the topcoat from containing 23% of the drug.
On appeal, Cordis argued that the district court erred by declining to construe the “non-thrombogenic” limitation to require less thrombogenicity than an uncoated metal stent. Cordis asserted that that Cypher stents and bare-metal stents were equally thrombogenic.
A little like politician, Cordis publicly said that its stents were non-thrombogenic, thus meeting the claim language and infringing, all while arguing that its public statements used the term in a different sense from the patent and that its stents do not infringe.
The Federal Circuit had their own thoughts:
To determine the meaning of “non-thrombogenic,” we begin by considering the language of the claims. See Phillips v. AWH Corp., 415 F.3d 1303, 1314 (Fed. Cir. 2005) (en banc). The language of claim 1 requires that the non-thrombogenic topcoat material “provide[] . . . non-thrombogenicity” to the stent, but it does not require a comparison to any other stent, either bare metal or coated. ’536 patent col.13 ll.22–26. As the claims themselves provide no other insight into the meaning of “non-thrombogenic,” we turn to the specification in order to determine if it provides a clearer indication of the scope of “non-thrombogenic.”
“[C]laims must be read in view of the specification, of which they are a part.” Phillips, 415 F.3d at 1315 (quotation marks omitted). The abstract of the invention states that the “non-thrombogenic surface . . . is provided with sites . . . which aid in . . . reduc[ing] thrombogenic activity.” ’536 patent abstract; see also id. at col.2 ll.36–46 (using heparin “to impart a non-thrombogenic surface to the material”). Thus, we can discern that the non-thrombogenic material must reduce thrombogenic activity because of its particular properties.
Cordis argued that the ’536 patent invalid for obviousness. According to Cordis, the Wolff patent alone renders the invention of claim 8 obvious since it shows a polymer stent made of a drug-eluting polymer with a barrier topcoat, and Wolff also refers to the stent and topcoat as separate “layers.” Cordis also argued that Domb alone also rendered it obvious because Domb teaches every limitation except that the stent can be made of metal and have an open lattice.
Boston Scientific countered that KSR is irrelevant to this obviousness inquiry because Cordis urged no particular combination of references. Also, had it been so obvious, Boston Scientific argues, Medtronic would have made the claimed invention.
Ultimately, the Court said the patent was dead:
We agree with Cordis that Wolff alone renders claim 8 of the ’536 patent obvious and therefore invalid. Because we hold the claim obvious based on Wolff alone, we do not address Cordis’s arguments for obviousness based on Domb or any other reference.
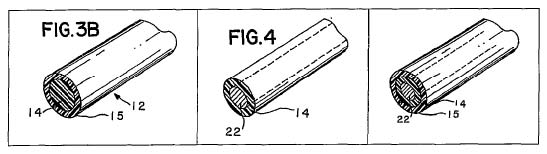
As we have discussed above, claim 8 recites a metallic stent with an open lattice structure. The stent includes an undercoat and a topcoat. The undercoat contains a drug, and the topcoat is drug-free and non-thrombogenic.
Wolff also discloses a stent including an undercoat and a topcoat, where the undercoat contains a drug. … Wolff also discloses that the topcoat is drug-free, as layer 15 in figure 3B “may be a simple barrier which limits diffusion of drugs” and “could be as simple as a silicone or polyurethane.” Wolff also discloses that the topcoat is non-thrombogenic.
Reversed.
Update: Boston Scientific and Medtronic agreed to settle two patent-infringement lawsuits over stents and put three others on hold. Boston Scientific said it will continue to pursue patent-infringement claims against Abbott Labs. It’s not personal, it’s just business.

I’d like to know what other readers think about Cordis’s disavowal of its public statements that tended to establish infringement, i.e., that Cypher was not thrombogenic. Smells bad to me. Like a patent, their statements are supposed to provide a type of notice to the public, no?
David French writes:
In Canada, a patent stands for what it is on its face. There can be no estoppel based on general statements made to the public, at least on the basis of patent law so far.
A dedication of the patent to the public or a declaration of a terminal disclaimer may qualify as an exception.
The Canadian courts want to avoid complicating patent cases with facts arising outside of the patent document itself and the prior art. There is no file wrapper estoppel in Canada. “Keep it simple” is the policy.
Is this fair? Well, at least it simplifies litigation.