The going has always been tough for getting pharmaceutical patents. The going is getting tougher. See Aventis Pharma Deutschland Gmbh and King Pharmaceuticals, Inc., v. Lupin, Ltd. (06-1530).
In a patent infringement action concerning the pharmaceutical compound ramipril, marketed by King as a blood pressure medication under the name Altace®, the U.S. Court of Appeals for the Federal Circuit followed the recent Supreme Court guidelines on obviousness and struck down a stereoisomer as obvious.
Lupin appealed from a judgment of infringement in which the district court concluded at summary judgment that Lupin’s filing of an Abbreviated New Drug Application (ANDA) for a generic version of ramipril infringed Aventis’s U.S. Patent. No. 5,061,722 under the doctrine of equivalents and concluding after a bench trial that the asserted claims of the ‘722 patent were not invalid.
On appeal, the Federal Circuit concluded that the subject matter of the asserted claims of the ‘722 patent would have been obvious.
The patent is directed to ramipril in a formulation “substantially free of other isomers.” Ramipril is one of a family of stereoisomers — an isomer in which the same atoms are bonded to the same other atoms, but where the configuration of those atoms in three dimensions differs. The following structural formula represents ramipril:
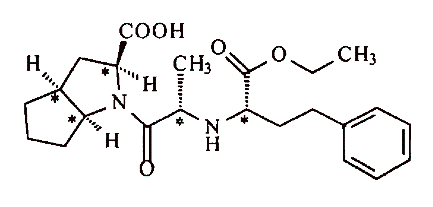
Because there are five carbon atoms that may take either of two orientations-or five “stereocenters,” as such atoms are known-ramipril is one of 25, or 32, stereoisomers. There are a number of different ways of naming these stereoisomers; one comparatively simple system, used by both parties and by the district court, involves labeling each stereocenter with an “R” or an “S” depending on its configuration. Using this system, all five stereocenters in ramipril are in the “S” configuration, so it is known as an “SSSSS” or “5(S)” stereoisomer. Other stereoisomers would include RRRRR, SSSSR, RRSSS, etc.
Some of the prior art references also use the terms “enantiomer” and “diastereomer.” Enantiomers are stereoisomers that are mirror images of each other, like left and right hands. Diastereomers are stereoisomers that are not enantiomers.
The asserted claims of the ‘722 patent read as follows:
1. A compound of the formula:
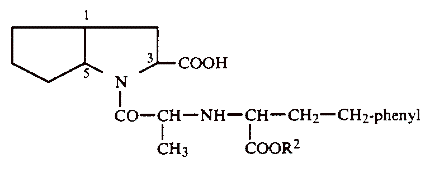
or a physiologically acceptable salt thereof, wherein R2 is hydrogen, methyl, ethyl, or benzyl, and wherein hydrogen atoms on the ring carbon atoms in the 1- and 5-positions are in the cis-configuration relative to one another, the carboxyl group on the ring carbon atom in the 3-position is in the endo position relative to the bicyclic ring system, and the chirality centers in the chain and on the ring carbon atom in the 3-position all have the S-configuration, said compound or salt being substantially free of other isomers.
Ramipril is one of a family of drugs known as “Angiotensin-Converting Enzyme inhibitors,” or “ACE inhibitors.” ACE inhibitors inhibit a biochemical pathway that constricts blood vessels and therefore are useful for treating high blood pressure.
Ramipril’s immediate predecessor is an ACE inhibitor known as enalapril. In a published article, Merck scientists explained that the all-S (SSS) stereoisomer of enalapril was found to have 700 times the potency of the SSR stereoisomer.
Soon after enalapril’s introduction, Dr. Elizabeth Smith, a chemist at Schering, conceived of the structure of ramipril and recorded it in her laboratory notebooks. Ramipril has the same overall structure as enalapril, with one distinction: where ramipril has two linked five-sided carbon rings (a “5,5 fused ring system”), depicted, in the chemical diagrams above, on the left side of the molecule, enalapril has only a single ring. The addition of the second ring gives rise to two more stereocenters than are present in enalapril; thus, ramipril has the same three stereocenters as enalapril, plus two new ones that span the fused ring system and are therefore known as “bridgehead” carbons, for a total of five as discussed above.
Schering obtained patents No. 4,587,258 and No. 5,348,944, which disclose the structure of ramipril but do not describe how its stereocenters should be configured. Example 20 of the ‘886 application discloses a method for making ramipril and yields a mixture of several, but not all, stereoisomers of ramipril, one of which is the 5(S) form.
In February 1981, Dr. Smith synthesized a mixture of 5(S)-configuration ramipril and its SSSSR stereoisomer. The district court concluded that Dr. Smith did not separate the 5(S) and SSSSR isomers, and there is no evidence that she conceived of a purified formulation containing only 5(S) ramipril.
The key question is whether the 5(S) stereoisomer of ramipril, in a form substantially free of other isomers, would have been obvious over the prior art still remaining “substantially free of other isomers.”
The district court held that Lupin failed to meet its burden of proof by clear and convincing evidence that a person of ordinary skill in the art would have been motivated to purify 5(S) ramipril into a composition substantially free of other isomers. The district court saw this as a close case based principally on the absence of a clear and convincing showing of motivation.
Since the date of that decision, however, the Supreme Court decided KSR International Co. v. Teleflex Inc., 127 S. Ct. 1727 (2007), which counsels against applying the “teaching, suggestion, or motivation” (“TSM”) test as a “rigid and mandatory formula[].”
The Federal Circuit noted that:
Requiring an explicit teaching to purify the 5(S) stereoisomer from a mixture in which it is the active ingredient is precisely the sort of rigid application of the TSM test that was criticized in KSR.
The “reason or motivation” need not be an explicit teaching that the claimed compound will have a particular utility; it is sufficient to show that the claimed and prior art compounds possess a “sufficiently close relationship . . . to create an expectation,” in light of the totality of the prior art, that the new compound will have “similar properties” to the old.
The analysis is similar where, as here, a claimed composition is a purified form of a mixture that existed in the prior art. Such a purified compound is not always prima facie obvious over the mixture; for example, it may not be known that the purified compound is present in or an active ingredient of the mixture, or the state of the art may be such that discovering how to perform the purification is an invention of patentable weight in itself. However, if it is known that some desirable property of a mixture derives in whole or in part from a particular one of its components, or if the prior art would provide a person of ordinary skill in the art with reason to believe that this is so, the purified compound is prima facie obvious over the mixture even without an explicit teaching that the ingredient should be concentrated or purified.
Ordinarily, one expects a concentrated or purified ingredient to retain the same properties it exhibited in a mixture, and for those properties to be amplified when the ingredient is concentrated or purified; isolation of interesting compounds is a mainstay of the chemist’s art. If it is known how to perform such an isolation, doing so “is likely the product not of innovation but of ordinary skill and common sense.”
The prior art supporting prima facie obviousness included the SCH 31925 mixture, and so Aventis must show that 5(S) ramipril had unexpected results not over all of its stereoisomers, but over that mixture, which did not contain the RRSSS form. And the potency of pure 5(S) ramipril is precisely what one would expect, as compared to a mixture containing other, inert or near-inert stereoisomers.

I find this extremely interesting – not least because I’ve spent years making drug candidates and worry about their patentability, and also because I used to work right down the hall from Liz Smith (!)
[…] The going has always been tough for getting pharmaceutical patents. The going is getting tougher. See Aventis Pharma Deutschland Gmbh and King Pharmaceuticals, Inc., v. Lupin, Ltd. (06-1530). In a patent infringement action concerning the pharmaceutical compound ramipril, marketed by King as a blood pressure medication under the name Altace, the U.S. Court of Appeals for the Federal Circuit followed the recent Supreme Court guidelines on obviousness and struck down a stereoisomer as obvious. source: Pharma Find Patents More Elusive for Stereoisomers, Patent Baristas […]
i need to know is their actual differences in potenct between 5 s & rrsss mixture kindly comment